Nitrogen: Difference between revisions
More actions
m →Sources: added source for nitrogen |
Added Phase Diagram |
||
| Line 12: | Line 12: | ||
| molar_mass = 64 g/mol <!--Mole:MolarMass, Chemistry.MOLAR_MASS_NITROGEN--> | | molar_mass = 64 g/mol <!--Mole:MolarMass, Chemistry.MOLAR_MASS_NITROGEN--> | ||
}} | }} | ||
[[File:Phase Diagram of Nitrogen.png|thumb|Phase Diagram of Nitrogen]] | |||
= Description = | = Description = | ||
| Line 21: | Line 23: | ||
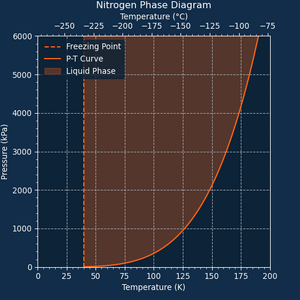
Nitrogen will condense into <b>Liquid Nitrogen</b> starting at {{Temperature|190}}. The boiling point of Nitrogen is {{Temperature|75}} at 100 kPa. | Nitrogen will condense into <b>Liquid Nitrogen</b> starting at {{Temperature|190}}. The boiling point of Nitrogen is {{Temperature|75}} at 100 kPa. | ||
Due to its high molar density, low heat capacity, and generally inert nature, Nitrogen is an excellent pressure agent for condensing other gases into liquids and keeping [[Water|water]] from evaporating passively. | Due to its high molar density, low heat capacity, and generally inert nature, Nitrogen is an excellent [[pressure]] agent for condensing other gases into liquids and keeping [[Water|water]] from evaporating passively. | ||
[[Soybean|Soybeans]] require Nitrogen to grow, but they will still exhale [[Oxygen]]. | [[Soybean|Soybeans]] require Nitrogen to grow, but they will still exhale [[Oxygen]]. | ||
| Line 52: | Line 54: | ||
| [[Ore_(Silver)|Silver Ore]] || 0.4 mol | | [[Ore_(Silver)|Silver Ore]] || 0.4 mol | ||
|} | |} | ||
producing Fertilizer in the [[Kit (Advanced Composter)|Advanced Composter]] | producing [[Fertilizer]] in the [[Kit (Advanced Composter)|Advanced Composter]] | ||
{{:Gases and Liquids/Menu}} | {{:Gases and Liquids/Menu}} | ||
[[Category:Gas]][[Category:Liquid]] | [[Category:Gas]][[Category:Liquid]] | ||
Latest revision as of 04:32, 26 March 2026
Description
Nitrogen can be mixed with Oxygen to stabilize its flammability and avoid Oxygen toxicity, and is a resource in its own right, useful for creating, a breathable atmosphere (at roughly 3:1 Nitrogen and Oxygen). The most efficient source of Nitrogen is from Ice (Nitrice) deposites found around the Solar System. You can Condense Nitrogen into Liquid Nitrogen by lowering its temperature below its boiling point.
- Stationpedia
Nitrogen, also referred to as N2, is an inert gas most often used in the creation of air.
Details
Nitrogen will condense into Liquid Nitrogen starting at 190 K (-83.1 °C). The boiling point of Nitrogen is 75 K (-198.1 °C) at 100 kPa.
Due to its high molar density, low heat capacity, and generally inert nature, Nitrogen is an excellent pressure agent for condensing other gases into liquids and keeping water from evaporating passively.
Soybeans require Nitrogen to grow, but they will still exhale Oxygen.
Nitrogen is used in the creation of Nitrous Oxide when combined with Oxygen in a Nitrolyzer.
Sources
You can collect Nitrogen from the following sources:
| Source | Amount |
|---|---|
| Ice (Nitrice) | 22.5 mol |
| Ice (Water) | 5 mol |
| Ice (Oxite) | 2.5 mol |
| Lead Ore | 1 mol |
| Silicon Ore | 1 mol |
| Nickel Ore | 0.5 mol |
| Gold Ore | 0.5 mol |
| Copper Ore | 0.5 mol |
| Silver Ore | 0.4 mol |
producing Fertilizer in the Advanced Composter
Gases and Liquids
| |||
|---|---|---|---|
| Elements | Compounds | Mixtures | Other |
|
|
|||
