Carbon Dioxide
More actions
Description
Carbon Dioxide, also referred to as CO2, is a non-flammable gas found in some atmospheres naturally (such as Mars). It can also be produced by burning Fuel or Coal, smelting most ores (e.g. Iron, Copper, and Silicon) and through human respiration.
Details
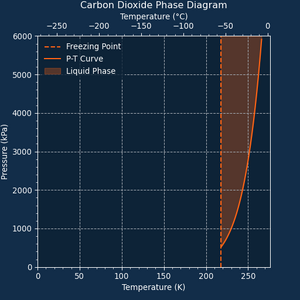
Carbon Dioxide will condense into Liquid Carbon Dioxide starting at 265 K (-8.1 °C). With such a low temperature requirement, most setups will store Carbon Dioxide as a gas and use Liquid Carbon Dioxide as a coolant.
Carbon Dioxide is required for most plants to grow. Plants will inhale the Carbon Dioxide to create Oxygen which can be harvested using vents. Stationeers should monitor their plants to ensure they always have enough Carbon Dioxide as, given enough time, they can convert all of it into Oxygen.
Carbon Dioxide has an isochoric molar heat capacity of 28.2 J/mol⋅K, making it a superior coolant to Pollutant outside applications involving phase change where Pollutant can make use of its greater latent heat capacity. Both are greatly surpassed by water, though water is harder to find and has more important uses.
Sources
You can collect Carbon Dioxide from the following sources:
| Source | Amount |
|---|---|
| Coal Ore | 10 mol |
| Charcoal | 3 mol |
| Copper Ore | 1 mol |
| Gold Ore | 0.2 mol |
| Iron Ore | 0.5 mol |
| Lead Ore | 0.4 mol |
| Nickel Ore | 1 mol |
| Silicon Ore | 1 mol |
Gases and Liquids
| |||
|---|---|---|---|
| Elements | Compounds | Mixtures | Other |
|
|
|||
