Hydrogen
More actions
Description
Hydrogen or H2 is a combustible gas. The main source of Hydrogen on most planets is in the form of Ice (Volatiles) that can be mined on the surface. Hydrogen will autoignite at 300°C or above in the presence of Oxygen, autoignite at 150°C in the presence of Ozone and autoignite at 50°C in the presence of Nitrous Oxide.
Combustion
Usage
See also: Fuel
While Hydrogen is a superior fuel for rocketry, there is currently no way to refine it from Methane, limiting its available supply. As such, it is recommended to combine it with Nitrous Oxide or Ozone in rocketry applications to maximize its effectiveness.
A suggested alternative use is as a working gas for a Stirling Engine, where it can achieve a conversion efficiency of 18%, surpassing the efficiency of Methane and tying it with Helium.
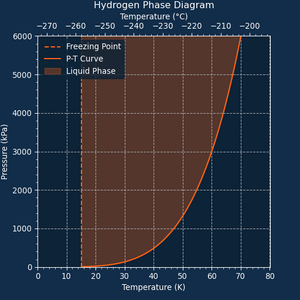
Hydrogen also has the lowest freezing point of all gases capable of phase change, making it the only choice for extreme cryocooling applications. However, its limited latent heat capacity hinders its performance in that task.
Notes
- The Specific heat of 20.4 Joule / mol is in-between the specific heat of Hydrogen and Methane in real-life.
- The actual Autoignition temperature of Hydrogen in Oxygen is approximately 570 °C at 1 atm, decreasing to 414°C at 7.4 MPa.
- Unlike Methane, Hydrogen is not toxic to most plants in-game.
- As far as rocketry is concerned, Hydrogen has a mass of 500 mol/kg (2 g/mol).
Sources
Hydrogen can be obtained by processing Water in an Electrolyzer or acquired from traders.
Additionally, Hydrogen can be collected from the following sources:
| Source | Amount |
|---|---|
| Ice (Volatiles) | 2 mol |
References
Gases and Liquids
| |||
|---|---|---|---|
| Elements | Compounds | Mixtures | Other |
|
|
|||
